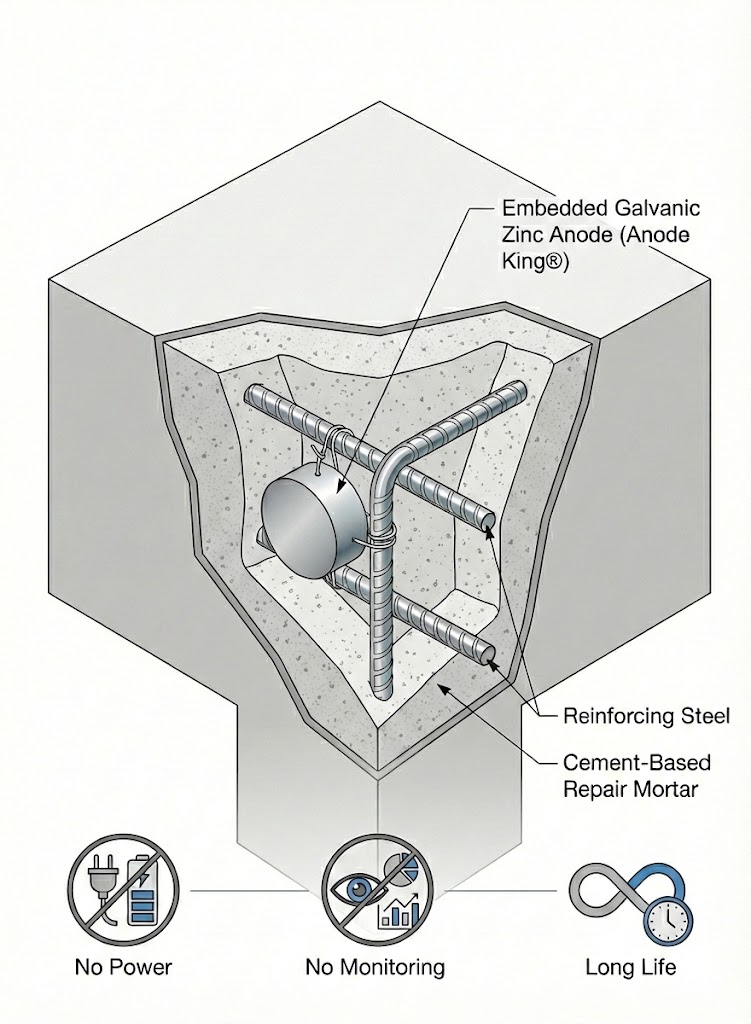
How Anode King® Works
Galvanic Corrosion Principle
When two dissimilar metals are electrically connected in an electrolyte, a galvanic cell is formed. In this system, the more electrochemically active metal acts as the sacrificial anode and corrodes preferentially, while the less active metal is protected.
This natural electrochemical process is the fundamental principle of galvanic corrosion protection, where controlled anode consumption is used to reduce or prevent corrosion of critical structural elements.
Anode King® is an embedded galvanic zinc anode designed to provide localized, maintenance-free corrosion protection for reinforcing steel.
Once installed, it operates autonomously through natural electrochemical processes, delivering reliable protection without the need for external power, monitoring, or ongoing maintenance.
Sacrificial Zinc Protection
In Anode King®, the zinc anode corrodes instead of reinforcing steel, reducing corrosion activity and protecting adjacent reinforcement.
Localized Protection Mechanism
Protection is focused at repair boundaries where corrosion risk is highest.
